Convection isn’t cool: battery thermal control during model development (Part 1 of 2)
- Kieran O'Regan and Christian Pilling and Jamie Higgins and Darryl Doyle

- Apr 11, 2024
- 7 min read
Updated: Jan 24

Understanding the impact of temperature on battery performance is critical for battery system development, but in most labs, this parameter is not controlled effectively. This two-part blog delves into the challenges of thermal control in battery model development. We will explore the reasons for these challenges, and more importantly how we solve them to produce high-quality data and parameters for models. The second part (released 17th April) will provide a detailed case study on the Molicel P45B (a cell we have reported on here and here), the high-power 21700 cylindrical cell commonly used in motorsports, aerospace and other performance industries.
Key Takeaways:
Challenges of Battery Testing: Characterising cell performance against temperature requires measurements to be taken at accurately controlled temperatures.
Benefits of Isothermal Testing: Our Peltier conductive thermal control method allows for precise temperature control of a cell under test.
Quantifying our Thermal Control Method: The Peltier system outperforms traditional methods by reducing temperature fluctuation by as much as 98%, resulting in more reliable battery test data.
The Status Quo
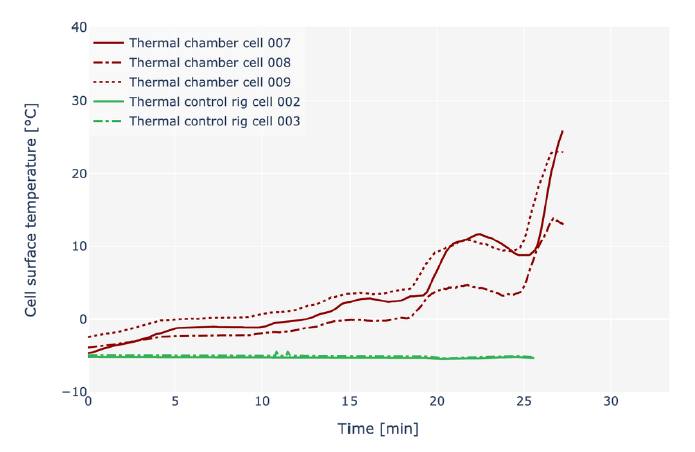
In industry, it is common to test lithium-ion batteries in thermal chambers powered by convection. This to control their temperature - or at least attempt to. However, the inherent limitations of convection mean that the cell temperature can drift significantly from the intended temperature set-point. This causes difficulties in the battery development process and ultimately the optimisation of electric vehicle performance. The graph below demonstrates how the Molicel P45B temperature increases significantly despite the best efforts of convection chambers to control this. Subjecting the P45B to a drive cycle sees it’s surface temperature rise by over 25 degrees in less than 30 minutes.
The issue around thermal control in battery R&D relates to a phenomenon known as “resistive heating”. This occurs when a battery generates internal heat during operation, particularly during charging and discharging cycles. Internal heating in batteries is primarily caused by resistances that convert electrical energy into heat during charging and discharging processes [1]. This heat generation increases under high current flows such as rapid charging, leading to elevated temperatures that convection alone is unable to control.

Significant temperature rise: The surface temperature of the Molicel P45B under a drive cycle using a convection chamber for thermal control.
The About:Energy Way
Our method of thermal control uses conductive cooling via Peltier elements, allowing us to strictly control the cell surface temperature, and offers a competitive edge in generating accurate battery data. Here, we demonstrate the benefits of our isothermal control method compared to traditional convection thermal chambers. The below graph shows the Molicel P45B under the same drive cycle as before, but this time its surface temperature is controlled to remain at a constant temperature. This enables us to make more meaningful correlation between battery performance in a lab to specific control points needed in pack and battery management system design.

Isothermal testing: The surface temperature of the Molicel P45B under a drive cycle using About:Energy’s thermal control.
Why is Accurate Temperature Control Important in Cell Testing?
Precise temperature control is crucial for creating accurate battery models and understanding degradation trends. We know that batteries are very sensitive to temperature, and often seek to characterise this relationship through measurement.
For a simple demonstration, let’s say that you want to measure how some cell quantity such as resistance changes with temperature. You might pick 5 different set temperatures, perform some characterisation cycle at each, and then extract the resistance from the measured voltage response. But if the cell-under-test isn’t adequately cooled, the actual temperature could increase relative to the intended temperature at each set-point.
The plot below uses an example dataset to illustrate how uncertainty in the temperature during measurement could result in a significantly different temperature-dependency trend of any measured quantity. The greatest errors are seen at low temperatures, where cell heating is greater.
Example measurement error: Demonstration of how a drift from the set-temperature when characterising a cell can result in a significantly different temperature-dependency trend. Set temperature being the thermal chamber temperature and Actual temperature being the temperature recorded on the cells surface.
This is of course a simple demonstration, and could be mitigated by using the directly measured temperature, or predicting the temperature change in the analysis stage using thermal modelling. However both add significant difficulty and uncertainty during analysis, for scenarios more complex than this example. Particularly for very complex tests such as degradation experiments, where the significant durations make it infeasible to account for temperature fluctuations.
Alternatively, experimental design could minimise the cell heating by using low currents and short durations. However such compromises are not always possible.
At About:Energy, our conductive thermal control opens up a wide range of dynamic cycles and high currents that we can use for parameterising battery models, whilst maintaining a more consistent temperature (reducing the electrical-thermal coupling effects that need to be accounted for in the analysis stage). This allows us to train our models under current duty cycles similar to what our customer’s see in a variety of real-world applications and separate electrical and thermal parameterisation into separate tasks, with improved accuracy in each.
Goldilocks Principle: Neither too hot nor too cold
Controlling temperature in lab experiments is important because it provides insights into how we should design batteries and how they would perform in the real-world. In an electric vehicle, especially in colder climates the higher cell resistances decreases with kinetic processes that occur within (lithium-ion transport) and lead to degradation. Higher temperature provide better kinetic, but this increased performance does not come without its problems, however; increased thermal energy speeds up not just the reactions we want, but side reactions that occur and degrade battery performance [2]. Electric vehicle makers need to balance this delicate interplay between performance and longevity:
Energy Capacity: Higher temperatures reduce internal resistance, enhancing lithium-ion transport and battery energy capacity.
Power Output: Elevated temperatures accelerate kinetic processes, increasing power output due to reduced resistance.
Battery Lifetime: Excessive temperatures hasten degradation, shortening battery life by increasing resistance and lowering cell capacity over time.

Goldilocks zone: The optimal operating temperature range of a battery balances performance and lifetime/safety. The actual range will vary considerably depending on battery chemistry and desired attributes in the application.
Finding this balance is where a thermal management system comes in. The thermal management system [3] in electric vehicles regulates the battery's temperature to ensure optimal performance and longevity, but it also consumes energy itself, making its efficiency crucial to overall vehicle performance. Now, with our method, we can precisely control parameters during battery pack development to better understand their impact on performance. This allows us to leverage data to make more informed decisions, significantly enhancing the quality and reliability of our battery systems.
Currently when batteries are tested in labs across the world - in automotive, aerospace, or manufacturing facilities - convection thermal chambers are commonplace. Under these conditions batteries can heat by as much as 50 °C during a test—this data can have utility for validation and real-world scenario testing. But throughout development isothermal data is crucial for training models used in EV and battery design, ensuring the models reflect real-world performance accurately.
Conduction vs. Convection
Traditionally, convection is the main mechanism used in thermal control with large thermal chambers, but it is not effective at mitigating the internal heating of batteries. Conduction is more effective at removing heat from a system and controlling battery temperature. Here the cell surface temperature is forced equal to the cooling plate set temperature, but in convection the surface temperature is free to differ from the chamber set temperature. This is where our innovative approach to battery testing comes in.

Conduction vs Convection: Illustration of convection, the process of heat transfer through fluid motion, and conduction, the direct heat transfer through material contact, highlighting the distinct mechanisms of thermal energy movement in each process. Area (A) through which heat is being transferred, higher temperature (T_H), lower temperature (T_C), thickness of the material (L), thermal conductivity of the material (k), heat transfer coefficient (h), and heat transfer rate (q).
About:Energy's unique thermal control method plays a critical role in industry research and development. Co-Founders Gavin White and Alastair Hales have developed, over several years, both the software and hardware for isothermal temperature control [4]. This hardware utilises conduction and the Peltier effect, a thermoelectric phenomenon where heat is pumped when electric current is passed. Use of thermoelectric devices enables enhanced thermal control to mitigate temperature increases in batteries during testing, and allows data acquisition at lower temperatures. This enables rapid changes in test temperature and facilitates "thermal drive cycles.”
Our thermal control rig employs copper for its excellent thermal conductivity, Peltier (thermoelectric) elements for precise temperature control, thermistors (as opposed to thermocouples) for accurate temperature sensing, and gold-plated connectors to ensure reliable and corrosion-resistant electrical connections. This combination of components provides a high-quality, low-cost testing setup for our laboratory. This setup can be adapted for various formats and sizes, and even allows pressure control for pouch and prismatic batteries.

Peltier Method: About:Energy's thermal control rigs for prismatic (left) and cylindrical (right) batteries. These adaptable rigs are designed to accommodate batteries of any size, providing high-quality data essential for informed engineering decisions. The schematic shows the Peltier elements, location of thermistor, and different electrical connectors.
There are commercial products that aim to achieve similar results, but are capital intensive and require ongoing investment into maintenance and personnel for years to come. About:Energy’s “The Voltt” provides access to leading hardware and process without the ongoing expense this would otherwise mean for a business (see our blog on the costs of battery test labs). Providing a path to “lab-less” battery technology companies obtaining high quality data at low cost. Improved data can be used to improve:
State-of-charge (SoC) Estimation: More accurate SoC estimation, optimising battery usage and energy efficiency.
Lifetime Prediction: Improved state of health estimations, enabling precise lifetime predictions and maintenance scheduling, reducing downtime.
Improving Thermal Management Efficiency: Ensures optimal performance and extending component lifespans, thus saving energy and costs.
Reducing Cost of Pack Designs: More efficient pack designs, leading to cost savings in materials and manufacturing processes.
Summary
Our thermal control units represent a significant advancement in experimental accuracy and efficiency. Our Peltier conductive thermal control method enables precise temperature regulation of a cell under test, significantly outperforming traditional methods with over 98% less temperature fluctuation and ensuring more reliable battery test data. This addresses the challenge of accurately characterising cell performance against temperature, which requires measurements at precisely controlled temperatures. The second part of the blog will explore advanced topics with the Molicel P45B, including state estimation, lifetime prediction, and thermal design, showcasing our method's business advantages. We remain dedicated to advancing and sharing our innovations in thermal control technology.





